
Building Laboratory Quality and Accreditation Systems
Summary: Worked with government and partners to implement a quality management system across tiered laboratories. Through SLIPTA mentorship and ISO accreditation support, Diag-Net helped clinics move from uncoordinated testing to accredited, reliable services—resulting in better patient outcomes and donor confidence.
Background:
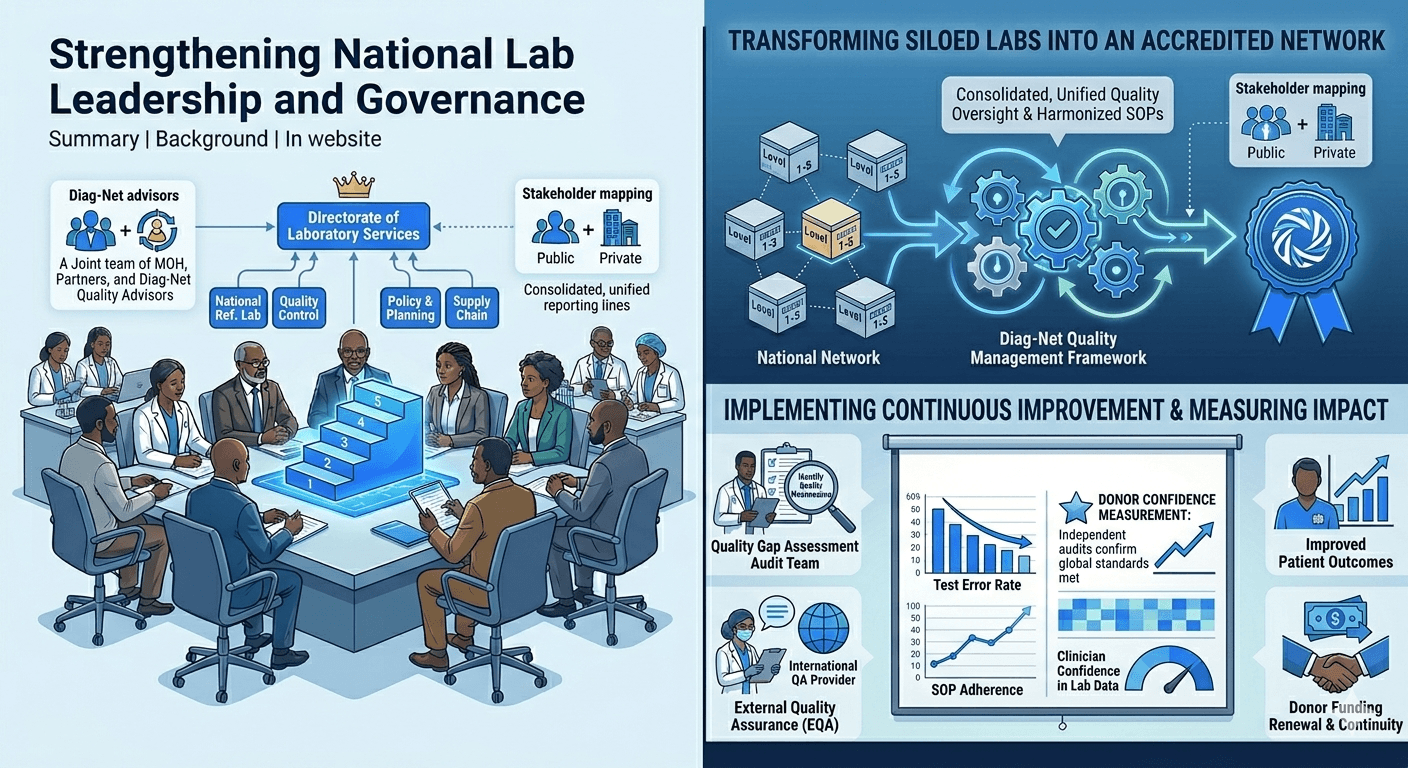
A national laboratory system lacked cohesive quality processes, leading to variable test accuracy. Diag-Net was engaged to develop a robust quality management framework across the lab network. We applied WHO-recommended SLIPTA and ISO approaches to drive continuous improvement[3].
Approach:
- Quality Gap Assessment: Performed baseline audits of key laboratories to identify quality weaknesses.
- SLIPTA & ISO Training: Implemented a stepwise training and mentorship program aligned with WHO’s SLIPTA standards [3].
- SOP Harmonization: Created standardized operating procedures for critical tests across labs.
- External QA Linkages: Established ties with international proficiency testing providers for ongoing assessment.
Outcomes:
Several regional labs achieved national accreditation readiness. The government noted a 30% increase in valid test results used for patient care. Donors renewed support when independent audits confirmed the labs met global standards. Clinicians reported greater confidence in lab data for treatment decisions, illustrating the impact of a structured Quality Management System.
