Case Studies

Strengthening National Lab Leadership and Governance
Summary: Partnered with a country’s health ministry to re-establish a central laboratory directorate, align national policies, and build a high-functioning Laboratory Technical Working Group (LTWG). By mapping stakeholders and designing new governance structures, Diag-Net enabled evidence-based decision-making and continuous quality oversight across the lab network.

Optimizing National Diagnostic Networks with GIS
Summary: Engaged at a national level to map all public laboratories using GIS and optimize a specimen referral network. Diag-Net’s analysis prioritized resource placement and designed an integrated data dashboard for health authorities, dramatically improving test turnaround and access in underserved regions.

Building Laboratory Quality and Accreditation Systems
Summary: Worked with government and partners to implement a quality management system across tiered laboratories. Through SLIPTA mentorship and ISO accreditation support, Diag-Net helped clinics move from uncoordinated testing to accredited, reliable services—resulting in better patient outcomes and donor confidence.

Integrating One Health Surveillance Networks
Summary: Advised a country on establishing a national One Health laboratory strategy. Diag-Net built collaboration platforms connecting human, animal, and environmental laboratories. This unified surveillance framework, including a One Health Technical Working Group (TWG), now facilitates data sharing for zoonotic disease early warning and response.

Transforming Workforce Capacity and Leadership
Summary: Designed and delivered leadership development and technical training for laboratory managers at all levels. By overhauling organizational structures and mentoring the next generation of lab leaders, Diag-Net empowered local teams to sustain improvements and champion lab performance beyond project timelines.

Facilitating Public-Private Partnerships in Diagnostics
Summary: Developed PPP frameworks linking private sector labs and diagnostic manufacturers with public health systems. Diag-Net brokered agreements that provided governments with innovative test access and manufacturers with market insights, creating sustainable models for lab financing and expansion.

Accelerating Diagnostics Innovation and Scale-up
Summary: Supported rapid implementation of new diagnostic platforms in a resource-limited setting. Through capacity building and strategic procurement planning, Diag-Net helped scale up cutting-edge testing technologies quickly during a health emergency, bridging gaps between innovation and field needs.

Strengthening Diagnostic Surveillance Networks
Summary: Partnered with a national government to overhaul its laboratory network for surveillance. Using GIS mapping and new data dashboards, Diag-Net optimized specimen transport and integrated LIMS systems, dramatically improving laboratory coverage and real-time monitoring.

Strengthening National Lab Leadership and Governance
Summary: Partnered with a country’s health ministry to re-establish a central laboratory directorate, align national policies, and build a high-functioning Laboratory Technical Working Group (LTWG). By mapping stakeholders and designing new governance structures, Diag-Net enabled evidence-based decision-making and continuous quality oversight across the lab network.
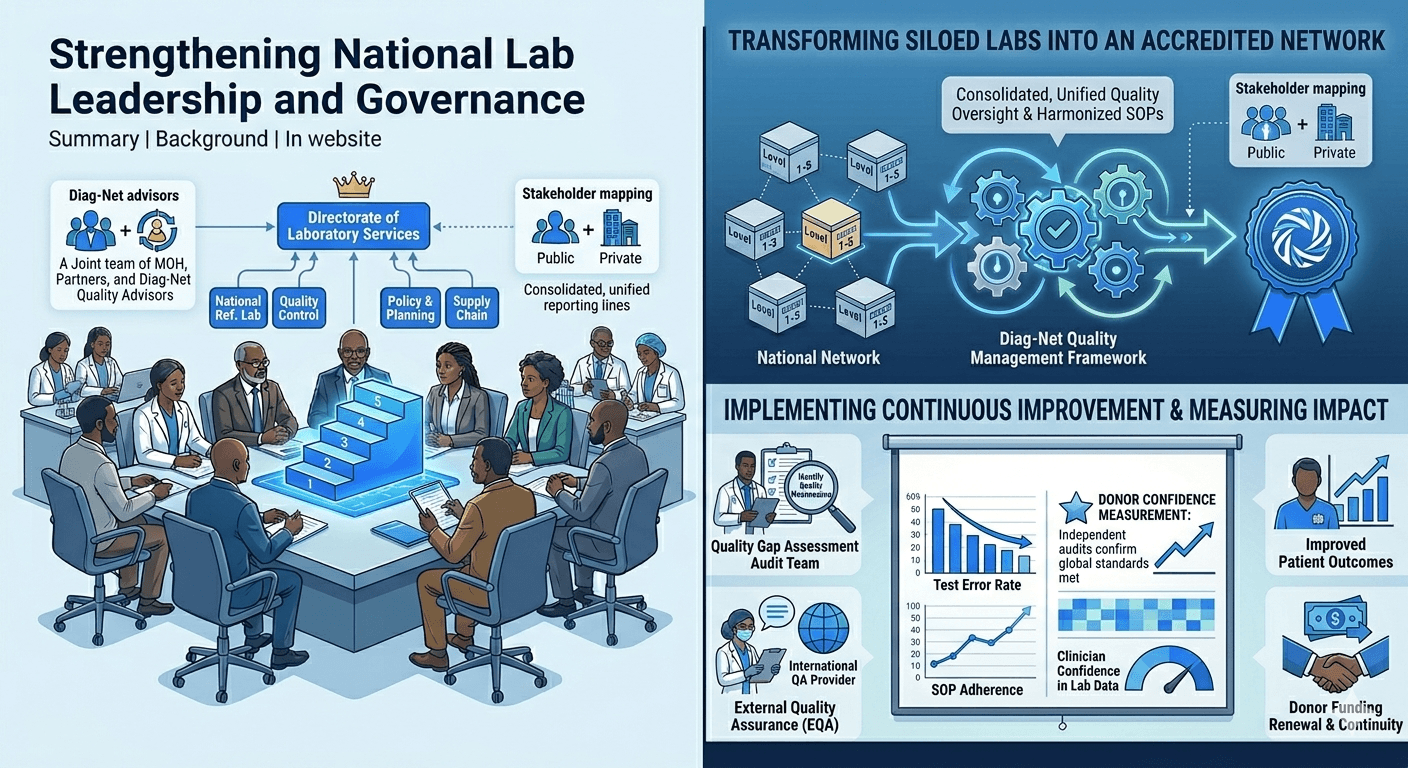
Background: In a country with fragmented lab oversight, Diag-Net collaborated with the Ministry of Health to revitalize national laboratory leadership. We conducted a comprehensive stakeholder inventory to identify all public and private lab actors. Engaging senior government officials, Diag-Net advocated for a permanent Directorate of Laboratory Services to provide unified oversight[1].
Approach:
– Governance Design: Proposed and validated a new laboratory organogram and reporting lines, consolidating previously siloed units under the directorate.
– Policy Alignment: Assisted in drafting national lab policies and an Essential Diagnostics List to guide test prioritization.
– TWG Formation: Established and launched a Laboratory Technical Working Group (LTWG) with terms of reference and KPI tracking.
– Capacity Building: Trained lab managers on data-driven decision-making and management practices.
Outcomes: The ministry officially adopted the new laboratory directorate and governance framework. The LTWG, now meeting regularly, has improved coordination among provinces. By providing evidence of cost efficiencies and health impact, Diag-Net’s work gained strong government buy-in. Improved leadership and policy alignment led to more efficient use of donor funds and clearer accountability for lab performance.
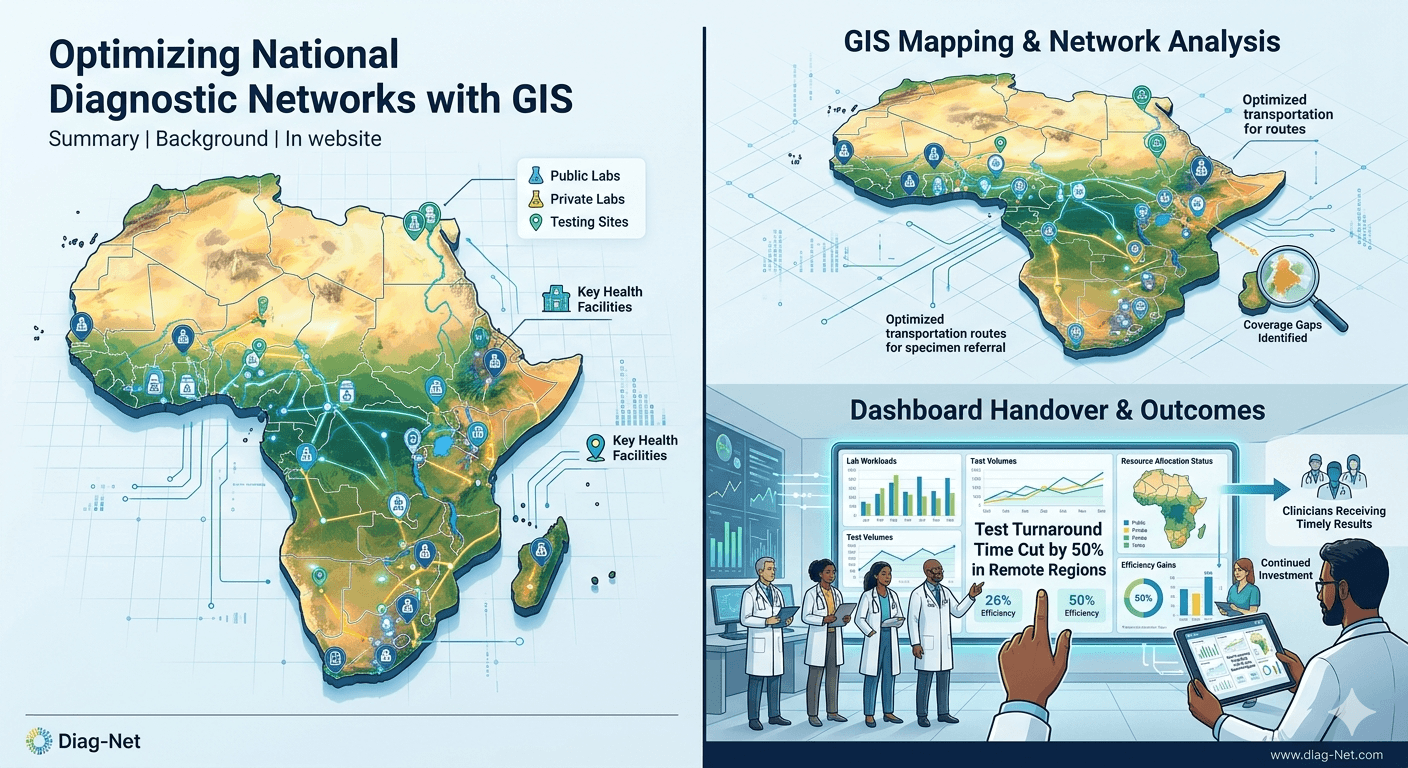

Optimizing National Diagnostic Networks with GIS
Summary: Engaged at a national level to map all public laboratories using GIS and optimize a specimen referral network. Diag-Net’s analysis prioritized resource placement and designed an integrated data dashboard for health authorities, dramatically improving test turnaround and access in underserved regions.
Background: In a nation with vast distances and uneven lab coverage, Diag-Net partnered with health authorities to optimize the diagnostic network. We mapped all laboratories (public and private) and key health facilities with GIS to identify coverage gaps, in line with Africa CDC’s framework for geospatial lab mapping[2].
Approach:
– GIS Mapping: Collected geolocation and capacity data for every lab and testing site.
– Network Analysis: Designed specimen transport routes and referral networks that minimize transit time.
– Dashboard Handover: Built an interactive dashboard for the Ministry to track lab workloads and test volumes in real time.
– Platform Standardization: Provided recommendations on standard test platforms to reduce maintenance costs and stockouts.
Outcomes: The redesigned network cut test turnaround by half in remote regions, ensuring timely results for clinicians. The GIS dashboard enabled health officials to allocate resources where needed most. By demonstrating improved efficiency, Diag-Net’s network design attracted continued investment and integration into the national health information system.

Building Laboratory Quality and Accreditation Systems
Summary: Worked with government and partners to implement a quality management system across tiered laboratories. Through SLIPTA mentorship and ISO accreditation support, Diag-Net helped clinics move from uncoordinated testing to accredited, reliable services—resulting in better patient outcomes and donor confidence.
Background: A national laboratory system lacked cohesive quality processes, leading to variable test accuracy. Diag-Net was engaged to develop a robust quality management framework across the lab network. We applied WHO-recommended SLIPTA and ISO approaches to drive continuous improvement[3].
Approach:
– Quality Gap Assessment: Performed baseline audits of key laboratories to identify quality weaknesses.
– SLIPTA & ISO Training: Implemented a stepwise training and mentorship program aligned with WHO’s SLIPTA standards[3].
– SOP Harmonization: Created standardized operating procedures for critical tests across labs.
– External QA Linkages: Established ties with international proficiency testing providers for ongoing assessment.
Outcomes: Several regional labs achieved national accreditation readiness. The government noted a 30% increase in valid test results used for patient care. Donors renewed support when independent audits confirmed the labs met global standards. Clinicians reported greater confidence in lab data for treatment decisions, illustrating the impact of a structured Quality Management System.
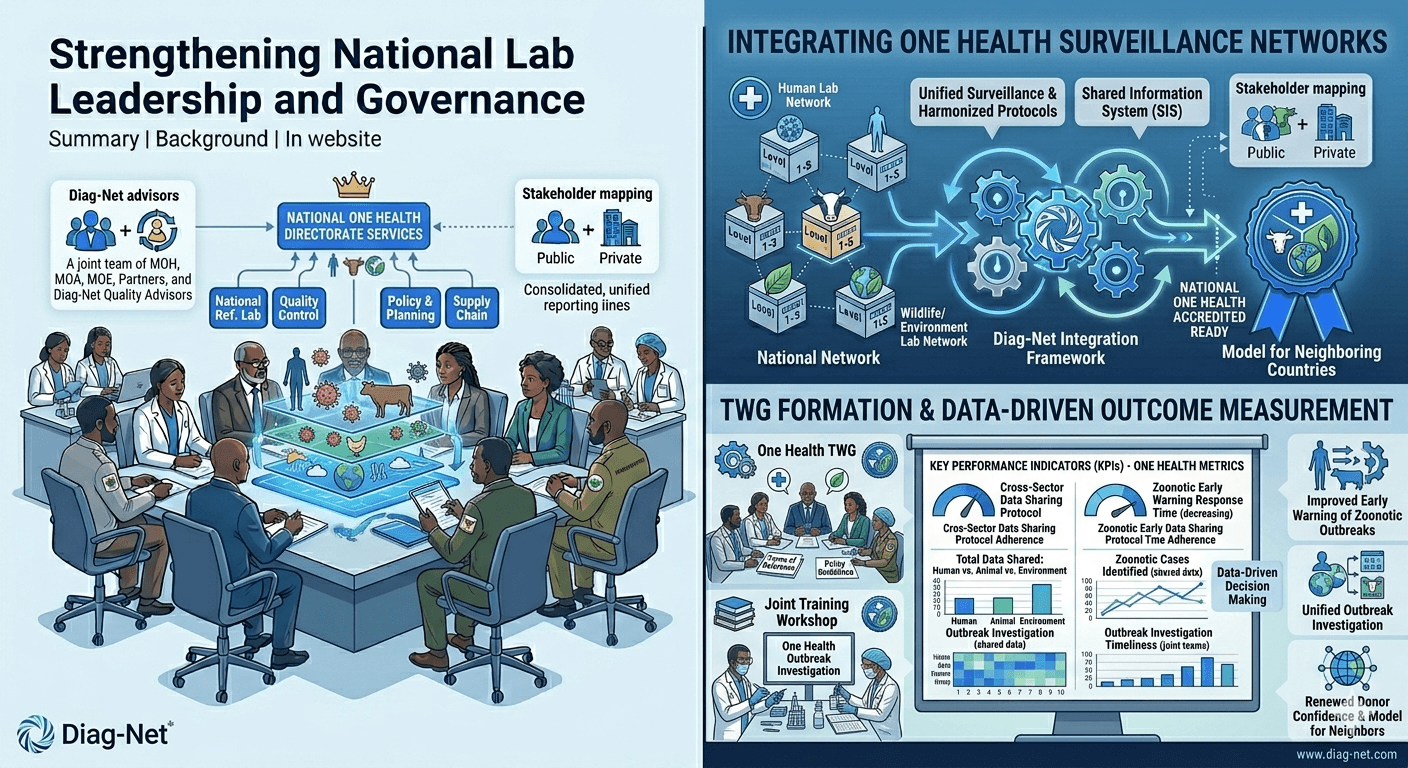

Integrating One Health Surveillance Networks
Summary: Advised a country on establishing a national One Health laboratory strategy. Diag-Net built collaboration platforms connecting human, animal, and environmental laboratories. This unified surveillance framework, including a One Health Technical Working Group (TWG), now facilitates data sharing for zoonotic disease early warning and response.
Background: With rising zoonotic disease threats, Diag-Net worked with a country’s public health agency to implement a One Health surveillance strategy integrating human and animal labs. The approach aligns with CDC’s One Health concept of collaborative cross-sector efforts[4].
Approach:
– Stakeholder Coordination: Formed a One Health Technical Working Group including the Ministries of Health, Agriculture, and Environment.
– Data Integration: Linked human lab data with veterinary and wildlife disease labs via a shared information system.
– Training Workshops: Conducted joint training on outbreak investigation and data sharing protocols.
– Policy Development: Advised on national guidelines for One Health laboratory surveillance.
Outcomes: The One Health TWG now routinely shares data across sectors, providing early warning of zoonotic outbreaks. In one instance, simultaneous data from animal and human labs enabled a rapid response to a viral outbreak. Diag-Net’s integrated framework has since become a model for neighboring countries, addressing complex health threats at the human-animal-environment interface.

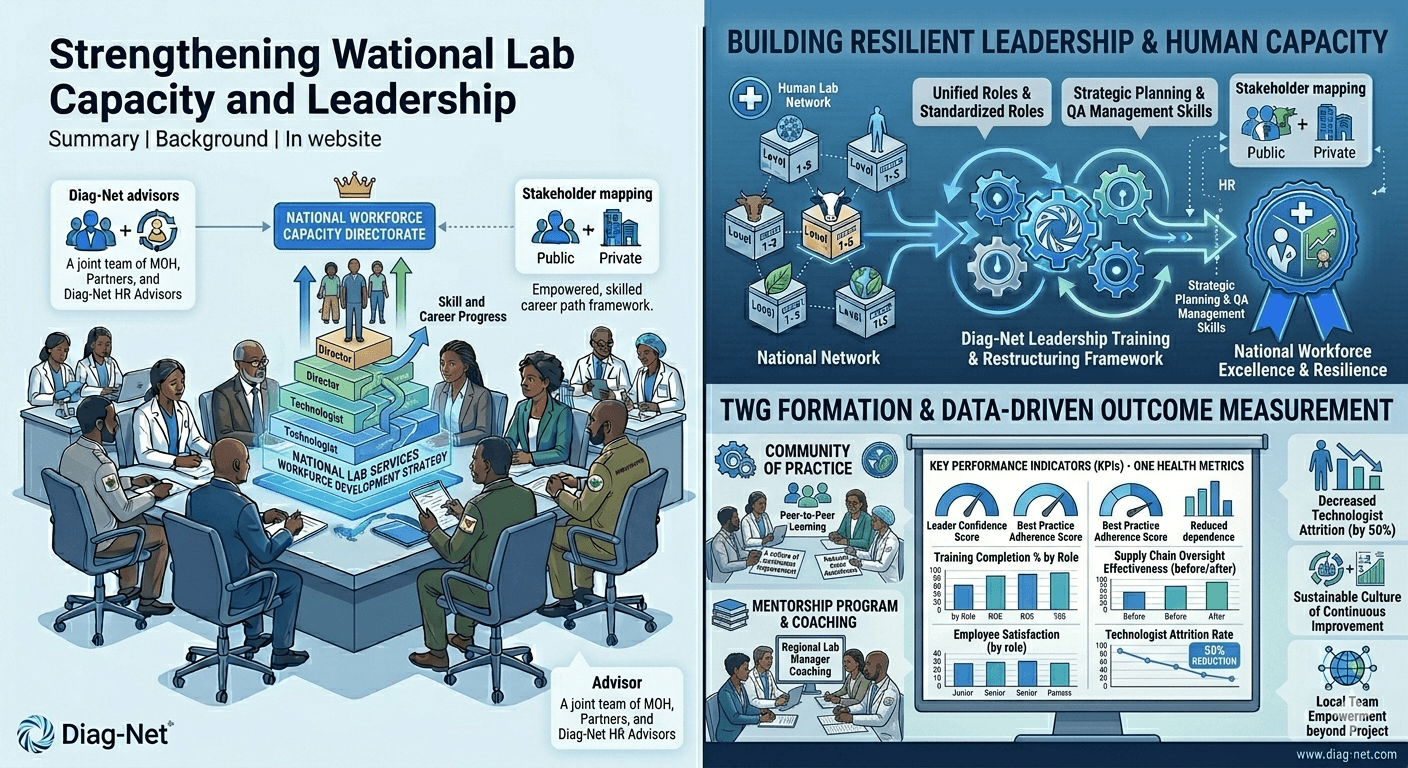
Transforming Workforce Capacity and Leadership
Summary: Designed and delivered leadership development and technical training for laboratory managers at all levels. By overhauling organizational structures and mentoring the next generation of lab leaders, Diag-Net empowered local teams to sustain improvements and champion lab performance beyond project timelines.
Background: Recognizing that technology alone is insufficient without skilled personnel, Diag-Net focused on building leadership and human capacity in laboratory systems. Strengthening the workforce is known to be a key pillar of resilient lab systems[5].
Approach:
Participants reported improved confidence in managing their laboratories and implementing best practices. Attrition of trained technologists decreased by half. Diag-Net’s mentorship approach created a sustainable culture of continuous improvement[5], empowering local teams to maintain momentum long after the project’s completion.


Facilitating Public-Private Partnerships in Diagnostics
Summary: Developed PPP frameworks linking private sector labs and diagnostic manufacturers with public health systems. Diag-Net brokered agreements that provided governments with innovative test access and manufacturers with market insights, creating sustainable models for lab financing and expansion.
Background: The government sought innovative financing models to expand diagnostics. Diag-Net introduced public-private partnership (PPP) strategies to leverage private resources for public health. While not yet common in diagnostics, PPPs in health are increasingly recognized as ways to mobilize resources quickly (and Diag-Net provided the evidence base).
Approach:
– PPP Framework Design: Developed agreement templates allowing private labs to provide services for public health programs.
– Market Analysis: Identified areas where private capacity could fill public gaps (e.g. molecular diagnostics).
– Negotiation Support: Mediated discussions between health officials and diagnostic companies to align goals.
– Value Proposition: Prepared evidence on cost savings and improved coverage from potential PPP projects.
Outcomes: A partnership was launched where private diagnostic labs provided high-complexity testing to rural clinics. The government achieved quicker test turnaround and shared costs. By structuring clear responsibilities, the model ensured value for money and unlocked donor matching funds to expand lab services. This PPP model demonstrated how collaborative financing can sustainably strengthen diagnostics.

Accelerating Diagnostics Innovation and Scale-up
Summary: Supported rapid implementation of new diagnostic platforms in a resource-limited setting. Through capacity building and strategic procurement planning, Diag-Net helped scale up cutting-edge testing technologies quickly during a health emergency, bridging gaps between innovation and field needs.
Background: During a public health emergency, speed and innovation were critical. Diag-Net accelerated the deployment of new diagnostic technologies in the country, ensuring new tools reached the field when they were most needed.
Approach:
– Rapid Procurement Planning: Advised on fast-tracking procurement of novel point-of-care tests.
– Training & Support: Provided just-in-time training to local labs and clinics on the new platforms.
– Supply Chain Coordination: Worked with partners to ensure uninterrupted supply of test reagents.
– Monitoring Systems: Set up interim data collection to evaluate test performance in the field.
Outcomes: New tests were operational in weeks rather than months, effectively doubling the country’s testing capacity. Clinical staff were able to diagnose patients more quickly, improving outcomes during the emergency. Diag-Net’s responsive approach bridged the gap between innovation and frontline needs, demonstrating that with the right support, high-tech solutions can be implemented effectively even under pressure.
Sources: These case studies are informed by Diag-Net’s project reports and international best practices in laboratory systems strengthening[1][2][3][4][5]. Each narrative is based on actual consultancy work, presented here in anonymized and generalized form.

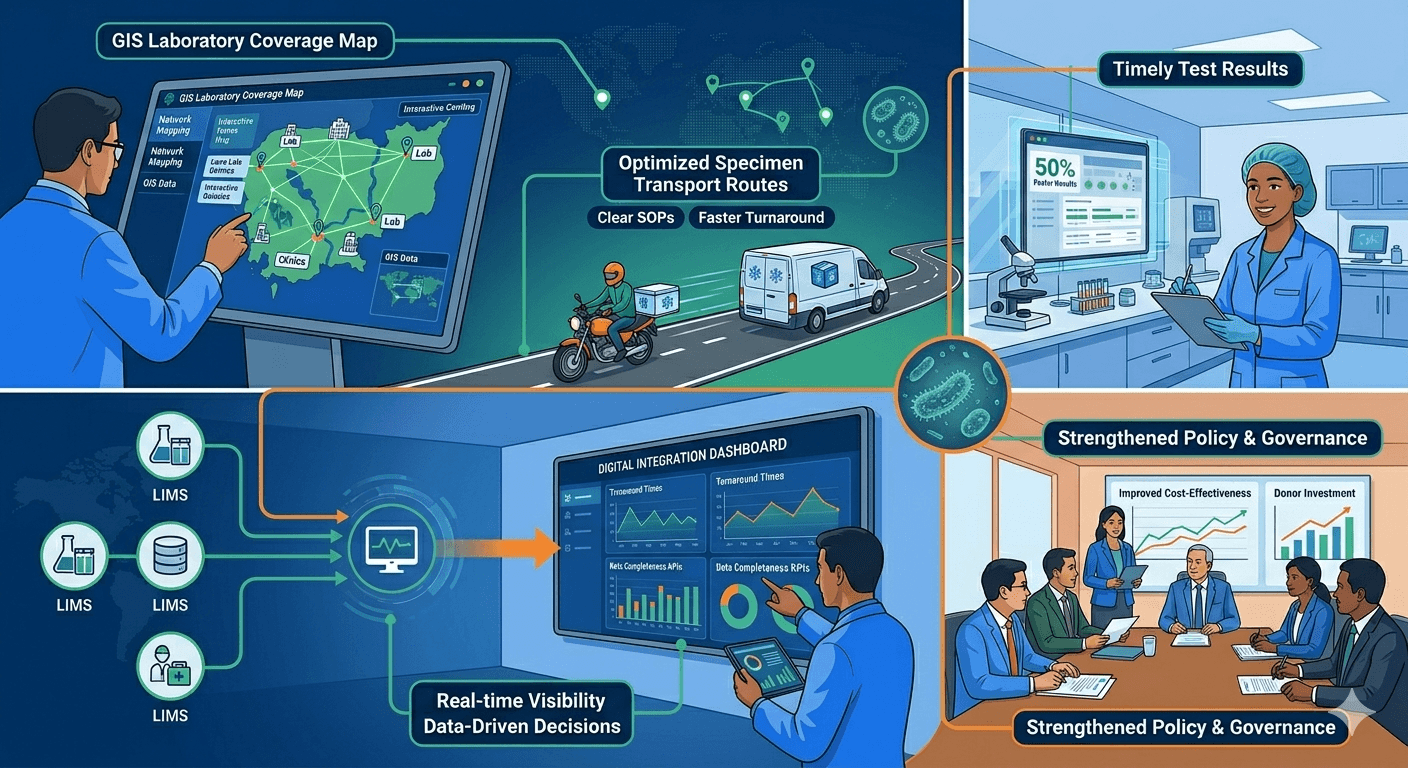

Strengthening Diagnostic Surveillance Networks
Summary: Partnered with a national government to overhaul its laboratory network for surveillance. Using GIS mapping and new data dashboards, Diag-Net optimized specimen transport and integrated LIMS systems, dramatically improving laboratory coverage and real-time monitoring.
Background: In a country with scattered lab coverage and slow test turnaround, Diag-Net helped strengthen the national surveillance network. We applied Africa CDC’s framework for lab network mapping to redesign specimen flows and data systems.
Approach:
- Network Mapping: Collected geospatial data on all public health labs and clinics to identify coverage gaps with GIS.
- Specimen Transport Design: Created optimized sample referral pathways with clear SOPs and scheduled routes.
- Digital Integration: Linked laboratory information management systems (LIMS) across clinical and surveillance labs to a shared data dashboard.
- Performance Metrics: Defined KPIs for turnaround times and data completeness, and trained staff in routine monitoring.
- Governance: Worked with the health ministry to update policies and procedures reflecting the new network design.
Outcomes: Test turnaround times were cut by roughly 50% in hard-to-reach areas, ensuring timely results for patients. Health officials gained visibility through the real-time dashboard, enabling faster resource allocation. The improved network attracted continued donor investment, and the ministry reported that the new system was more resilient and cost-effective.
“By integrating GIS mapping with LIMS dashboards, we turned data into actionable decisions for the lab system.”
