You may remember standing in a busy health centre, waiting while a nurse or doctor printed your test results. In a few seconds, those results could change your life—or your child’s. You trusted them because people’s lives depend on the accuracy of the numbers on that slip of paper. Only later might you wonder how certain we can be that this result is correct, whether it comes from a village clinic or a private hospital in Europe. For millions of families worldwide, that quiet moment with a Laboratory report is the frontline of the health system.
Why Laboratory Quality Matters
Laboratory results drive critical decisions: starting or stopping a treatment, detecting an infection, or reassuring a worried family. When test quality falters, the consequences can ripple through the health system. Misdiagnoses lead to wrong treatments and avoidable harm, and clinicians lose trust in diagnostic services. In fact, studies in Africa have linked poor access to quality Laboratory testing with increased morbidity, inappropriate drug use, and loss of life [5]. Globally, the picture is stark: nearly half the world still has no reliable access to diagnostics [4]. In many countries in Africa, only about 30% of clinics have the basic equipment and supplies needed for common tests [4]. In short, quality is the bedrock of effective healthcare, from treating any disease to controlling outbreaks. Every diagnostic success begins with reliable Laboratory processes, no matter where they occur.
Bridging Past & Present
These milestones mattered for individual laboratories, but new challenges showed that countrywide strategies were needed. The HIV epidemic taught us the power of scaled diagnostics. Later, Ebola outbreaks revealed how weak laboratory networks can hamper testing across regions [8][9]. Pandemic threats such as COVID-19 proved that no single reference laboratory can keep a whole country safe — coordinated national systems are what make the difference.
Global health programs then developed stepwise approaches. In 2009, WHO AFRO launched the Stepwise Laboratory Quality Improvement Process Towards Accreditation (SLIPTA) in Kigali — a graded, star-based audit process to help laboratories move toward ISO 15189 — paired with the Strengthening Laboratory Management Toward Accreditation (SLMTA) training program, which taught laboratories how to improve. These programs showed it was possible to move many laboratories forward together.
At the same time, and responding to a call from the Global Health Security Agenda [7], experts began to focus on systems, not just sites. The LABNET scorecard introduction manuscript paper [8] noted that; there were no simple indicators for national Laboratory network functions. In outbreak settings, this gap limited countries’ ability to quickly identify weaknesses and respond effectively. In past outbreaks (like West African Ebola), this lack of system indicators hampered response [5]. IN 2015, the LABNET scorecard provided laboratory network metrics to complement WHO’s Joint External Evaluation (JEE) process for country readiness. This shift in thinking was critical: laboratory networks and the system underlying them are essential not only for healthcare delivery but also for preparedness, outbreak response, and Global Health Security (GHS). Strengthening laboratories at national scale and be able to measure it became a clear priority.
Put simply, the global health community moved from asking “Is this test result valid?”; “Is this laboratory good?” to “Is our country’s diagnostic systems strong?” The next step is connecting those dots: from test result/facility quality and standardization to mature national systems.
Introducing Laboratory Systems Maturity Monitoring (LMM)
If SLIPTA and ISO 15189 helped us improve individual laboratories, Laboratory Systems Maturity Monitoring (LMM) helps countries assess whether the whole system is ready to provide high-quality diagnostic services to the population. LMM enter as a logical next powerful tool in the continuum. LMM is a structured national-level assessment framework: it measures a country’s entire Laboratory system capability against goals like Universal Health Coverage (UHC), International Health Regulations (IHR) and Global Health Security (GHS). LMM provides an updated framework to measure the maturity of laboratory system towards emerging priorities such as integration, self-sufficiency, cost-effectiveness and community centered functions.
It is important to distinguish between a laboratory system and a laboratory network, because LMM is concerned with both. A laboratory system is the broader national health system structure that provides the governance, financing, workforce, policies, infrastructure, quality frameworks, data systems, and other core functions needed to make laboratory services sustainable and effective, whereas a laboratory network is the organized connection of laboratories across the different tiers of the health system, working together at national and wider levels to deliver clinical testing, support surveillance, monitor quality, and detect and respond to public health events.
In practice, LMM is a national-level assessment tool, developed by Africa CDC and WHO AFRO in collaboration with global partners. Instead of asking whether a single Laboratory is high quality, it looks at the whole system: governance and financing, workforce, supply chains, diagnostic networks, quality management, biosafety, data, infection prevention and control, and how laboratories connect to disease surveillance. Using a five-step maturity scale—from “no system in place” to “sustainable system”—countries can see where they stand and what to fix first [6].
Why LMM? Because by this point, the goal is to evolve beyond fixing one Laboratory at a time, but to strengthen the whole system. The Africa CDC/WHO AFRO LMM package explains that the assessment helps countries pinpoint gaps in their national Laboratory system, set priorities, and justify investment to improve diagnostic services for all [6]. For example, in one assessment, countries have used LMM findings to lobby for Laboratory budgets and update national Laboratory strategic plans. Importantly, LMM is meant to be repeated every few years to track progress over time and adjust improvement efforts as needed.
LMM in Practice:
- Collaborative, multi-day assessment (country-led committee + external facilitators)
- Identifies system weaknesses (e.g. lack of biosecurity plan, data reporting gaps, workforce issues)
- Generates a report for countries to create action plans and prioritize investment to strengthen diagnostic systems at the nationallevel [6]
Think of LMM as the next chapter after quality and accreditation: it focuses on national system maturity. It’s like moving from checking that each player on a team is skilled to measuring how well the whole team plays together.
The Laboratory Quality Continuum
One helpful way to understand this journey is to see laboratory quality not as a single event, but as a continuum.
It begins with reliable test results at the bench and builds upward through each level of the system, eventually linking to Universal Health Coverage (UHC), the International Health Regulations (IHR), and Global Health Security (GHS). In other words, we go from “does this test work?” to “does the whole system work together to keep everyone healthy?”
- Internal Quality Control (IQC):This is the starting point. It asks whether each individual laboratory test is accurate and precise. We check this with daily internal QC procedures for every run.
- External Quality Assessment (EQA):The next layer asks whether the entire testing process is consistent and reliable over time and across all laboratories. We evaluate this by participating in external proficiency testing (EQA) programs for each test.
- Quality Management System (QMS) Implementation:Beyond individual tests, a laboratory must have a strong quality system in place. This step assesses how well all elements of the QMS are implemented in the laboratory (it’s test-agnostic, covering every process). Structured programs like SLIPTA and SLMTA are used here.
- Accreditation (ISO 15189):Now we formally assess the lab’s competence. This checks whether the laboratory can perform complete medical testing processes in compliance with ISO 15189 standards. Accreditation attests that the Laboratory consistently meets those requirements.
- Laboratory Network Functionality:. Here we examine how well the network operates — for example, specimen referrals, coordination, and support structures. This is evaluated as part of the LMM process at the network/system level.
- National Laboratory System Maturity (LMM):The focus shifts from individual laboratory facilities to the network of laboratories and the underlying systems across the country. The LMM evaluates the entire national laboratory system. It allows us to measure how well governance, policies, financing, workforce, and information systems support national health goals, the Universal Health Coverage (UHC) and the International Health Regulation (IHR).
- Joint External Evaluation (JEE) and Global Health Security (GHS): This is the highest level of the continuum. Here, the focus shifts from laboratory system performance to how well the national health system can prevent, detect, and respond to public health threats. The JEE measures country capacity under IHR, including key laboratory system-related functions. In this sense, the LMM helps strengthen the laboratory system capacities that support better JEE outcomes, improved IHR compliance, and ultimately stronger global health security.
Together, these elements form a continuous pathway of quality, and Laboratory Systems Maturity Monitoring (LMM) is the lens that brings the whole journey into focus. It does not replace quality control, external quality assessment, quality management systems, or accreditation; it builds on them to determine whether the national laboratory system is strong enough to deliver equitable diagnostics, advance Universal Health Coverage and the International Health Regulations, and support global health security.
At this stage, quality is no longer judged only by the correctness of a result or the accreditation of a laboratory, but by the maturity, coherence, and resilience of the system that connects them all.

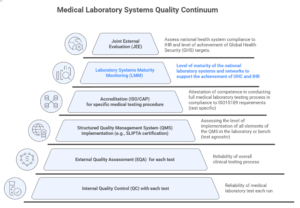
Figure: Imagine each step as a platform building to the next, from a bench in a community clinic up to global health security (from a QC to GHS).
Bottom Line
Quality is a journey, not a moment. Every laboratory result still starts with a technician trusting the controls, but we no longer stop there. We moved through accreditation to international standards, and now we are lifting our eyes to whole countries’ diagnostic capabilities. LMM is the culmination of that journey — a way to turn laboratory quality into countrywide capacity. By methodically measuring national Laboratory systems, LMM helps ensure investments and policies translate into reliable diagnostics for every patient and every outbreak. In doing so, it helps improve JEE performance and strengthen global health security, not just fix one gap.
Call to Action
Think of one aspect of your own national Laboratory system: a process or policy you’d like to “mature.” How could a national Laboratory system maturity assessment help? We encourage Laboratory leaders and policymakers to reflect on their quality journey. If you have examples or questions, please share them in the comments below. In the next LMM Series article, this series will rewind to the early days of accreditation and regulation—how tools like CLIA, CAP, and ISO 15189 first put structure around Laboratory quality, and what lessons they hold for countries now building whole Laboratory systems. Stay tuned.
======================================================================================================================
P.S., LMM is a national-level assessment tool, developed by Africa CDC and WHO AFRO, with the technical coordination of DATOS and support from the Global Fund. The tool benefitted from the high level of expertise and experience of the WHO Center of excellence for public health laboratories, The Clinton Foundation, The American Public health Laboratories (APHL), FIND, the Fondation Mérieux, ASLM, UNITAID, Matahari, the US government and various directors of national laboratory services of the WHO-AFRO region.
To learn more about LMM visit link: Africa CDC LMM Home
======================================================================================================================
References:
- College of American Pathologists, Historical Timeline of CAP Accreditation, January 1964 (CAP official website).
- U.S. Centers for Medicare & Medicaid Services, Clinical Laboratory Improvement Amendments (CLIA) fact sheet, 1988 (CMS.gov).
- International Organization for Standardization, ISO 15189:2003 Medical laboratories – Requirements for quality and competence, 2003.
- World Health Organization, Regional Strategy on Diagnostic and Laboratory Services and Systems 2023–2032 for the WHO African Region (WHO AFRO, 2023).
- Onyango, E.N. et al., “Building National Laboratory Systems in Africa: Aligning with Global Health Security Agenda,” African Journal of Laboratory Medicine, 2016.
- Africa CDC / WHO Regional Office for Africa, Laboratory Systems Maturity Monitoring (LMM) Country Guide 1.0, Dec 2025.
- Nkengasong and Skaggs. Are post Ebola reconstruction efforts neglecting public health laboratory systems. The Lancet Global Health;3(11):e678. 2015
- P. Ondoa, et.al, A new matrix for scoring the functionality of national laboratory networks in Africa: introducing the LABNET scorecard. Afr J Lab Med. 2016 Oct 3
- Cancedda C, et al. A comprehensive district-level laboratory intervention after the Ebola epidemic in Sierra Leone. African Journal of Laboratory Medicine. 2019